Please try these at home

Parts for the water pump:
- one or two AA batteries,
- a small plastic lid to hold a little water,
- a short length of copper wire,
- a short length of sticky-back copper tape (available from garden centres), and
- several small circular metal-coated magnets (available from online retailers).
Step 1:
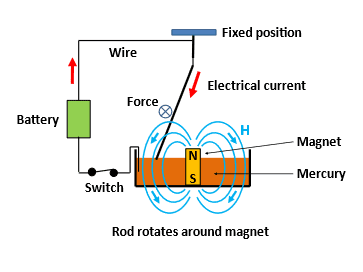
The idea for the water pump occurred to me when I was thinking about Faraday’s motor, so it is helpful to describe Faraday’s apparatus briefly. A sketch of his set-up is shown in Fig. 2.
Faraday placed a bar magnet vertically in a dish of mercury. A metal rod was suspended above the magnet from a fixed position with a piece of wire and the other end touched the surface of the mercury. Therefore, the rod was free to move around the point of suspension. A battery (a “Voltaic pile”) was connected to the fixed point above the magnet and, through a switch, to a wire that dipped into the mercury. When the switch was closed, current (red arrows) would flow from the battery down through the rod, through the mercury and back to the battery. Some components of the magnetic field of the magnet are perpendicular to the current in the rod, and therefore a force was generated perpendicular to both the current and the magnetic field. Consequently the rod rotated around the magnet. Faraday demonstrated this in 1821. It was the first time anyone had shown the conversion of electrical energy into mechanical energy was possible, and now, 200 years later, electrical motors are everywhere from laptops to domestic appliances to power stations.
Step 2:
In thinking about this experiment, I wondered “What would happen if the rod were held stationary?” The current and magnetic field are still there, so the force is still there. The force must now act on the mercury because that is the only moveable part. If that were the case, in principle, the mercury would be pushed around instead of the rod. I do not know if Faraday tried holding the rod still, but if he had done so I do not think anything noticeable would have happened because the mercury would have been too heavy to move. I doubt the current and magnetic field available to Faraday would have been strong enough to produce any detectable movement of the dense liquid metal.
So I thought I would check my thinking with a little experiment using a lighter, less dense, liquid, i.e. water. Water is about fourteen times less dense than mercury.
Step 3:
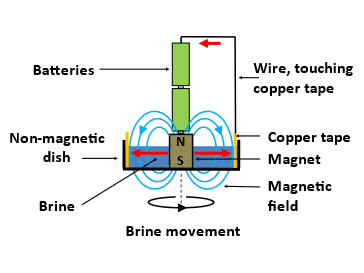
The simple water pump shown in Fig. 3 has a shallow non-magnetic dish (I used a plastic lid about 5 cm in diameter) containing a 1-2 millimetres depth of water. The arrangement sits on a tabletop. In the centre of the dish is a ceramic magnet that provides a vertical component of the magnetic field in the water. The “magnet” is a stack of five disc-shaped magnets, each one of which has a diameter of 12 mm and a thickness of 2 mm and a metal coating. [These are readily available online.] A strip of sticky-back copper tape (or bare copper wire) is placed around the inside edge of the dish in the water to constitute a second electrode. The AA batteries stick to the magnet. When the wire is held in contact with the top of the two batteries, and the other end is touched on the copper tape, electrical current (shown by red arrows) flows through the water from the surface of the magnet to the copper tape. The current and magnetic field are perpendicular to each other and consequently, a force is generated in the water that is perpendicular to both the current and magnetic field, i.e. circumferentially. This force pushes the water around the magnet. The movement is more easily seen if some small objects are floating on the water. I used very small pieces of a leaf. If the water does not move, try reducing the volume of water, e.g. reducing the depth from 2mm to 1mm. The flow is not fast, but it can be seen clearly.
Lesson of the Day
The speed of the water depends upon the strength of the force, which depends on the strength of the both the current and the magnetic field. The current will depend on the conductivity of the water. The higher the conductivity, the higher the current. Pure water has a low conductivity [4 x 10-5 S/m], drinking water has a higher conductivity [5 x 10-3 S/m], and sea water has a conductivity higher still [5 S/m]. So it is useful to start with tap water, and then add a few grains of table salt if necessary. Another variable is the voltage. I have shown two batteries, but more can be used although the structure may become unsteady.
The fact that the water moves shows that, in principle, a ship could be propelled using this idea, but I doubt it would be an efficient system. However, the idea might be useful in fluidics (fluid logic) or in nanotechnology for the remote control of internal drug delivery.
This DIY Guide is prepared by Professor Davis

Professor Lionel E. Davis wrote simple experiments (like this one) for his children and grandchildren. He used to be a communications engineer and educator. He aspires for all children to learn meaningfully at home. Prof. Davis is also one of the big motivators behind our work at Chumbaka and DreamCatcher. He has always inspired us to be more involved in Engineering & Radio Frequency. He is a Fellow at the Institute of Electrical and Electronics Engineers (IEEE) and the Institute of Physics (IOP).
